Learning with McCell - part 7: Iodine number
Ready for a new lesson with McCell? Today he will tell us about the Iodine number.
The Kaufmann method to measure the iodine number is used to characterize fats and oils with a visual titration. The iodine value indicates the quantity of unsaturated bonds in a fat molecule. With this measurement, the amount of iodine in grams which can bind to 100g of the fat is determined.

We can use the procedure to identify the fat and test its purity, because every fat or oil has a specific iodine number. It can also be used to make a statement about the quality of the fat, when you determine the number and compare it to already existing literature.
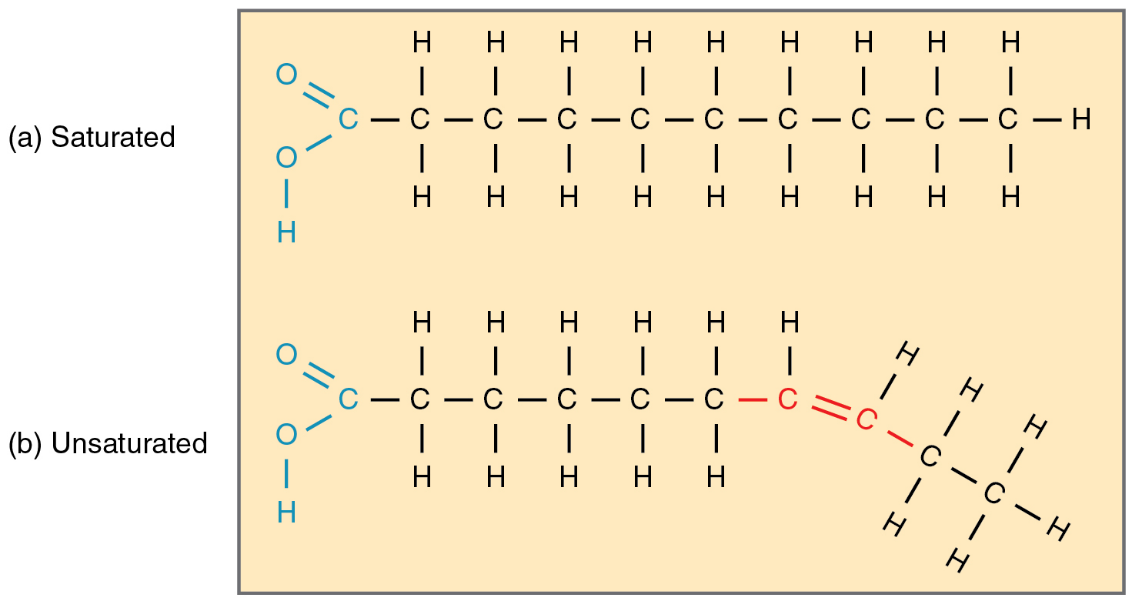
Fats (for example butter) can get rancid, if they are exposed to UV-light, oxygen, humidity as well as enzymes and microscopic organisms. These factors can dissect the fats which can lead to “degradation products”. Degradation products do not only smell bad, they can also harm your health. The two weak points of a fat are the ester bonds and the double bonds (C=C). Some weak points can easily get attacked by oxygen (O), enzymes and water. This process can lead to a transformation from healthier unsaturated fats to unhealthier saturated fats. One speaks of a saturated fat if the carbon atoms are only connected once (C-C). On the other hand, one speaks of an unsaturated fat, if the carbon atoms are bonded twice (C=C).
The initial reaction is performed with bromine and afterwards converted into iodine. Bromine is added in excess and the unused amount of it which is converted to iodine gets measured. First a small part of the sample gets weighed out. Afterward chloroform (to dissolve the fat) and bromine solution are added. The solution turns yellow or orange. Then the composition is closed and stored in a dark place for 2 hours where the reaction takes place. During this reaction the bromine splits the double bonds (C=C) to create single bonds (C-C). The sample is stored in a dark place to prevent other possible reactions that could take place. Then a potassium iodide solution is added and the whole sample is mixed well. The iodide in the solution reduces the bromine that wasn’t used for the reaction to bromide and is oxidated to iodine. Because of the iodine the mixture turns brown. The colored mixture is titrated to a transparent mixture with a sodium thiosulfate solution to determine the amount of iodine that has formed. To calculate the iodine number, we mix a “blank sample”, which contains the same ingredients as the sample, except the fat which one wants to measure. The result of the blank sample gets subtracted from the sample at the end. Afterwards one uses the previous result to calculate the iodine. This result is what we call the iodine number, which has the unit ml/g of fat.








